The nonionic detergents are the type of cleaning agent used in the pharmaceutical industry as it helps sanitize and protect the equipment in order to prevent poor quality and thus uphold the quality of the product.
Different Preservatives And Concentration Dosage For Pharmaceutical Liquid Preparation
As companies move to clearer labelling, LFA Machines is observing less and less use of chemicals in its customers’ products. Nevertheless when wet granulation is employed, for example, it is sometimes impossible not to have to include a preservative in the product formulation.
In the pharmaceutical industry, many of the ingredients used in formulations have the ability to promote microbial growth. Such formulations are therefore susceptible to unwanted contamination. To prevent this from occurring, antimicrobial agents or preservatives need to be added to the formulation.
In concept such preservatives protect the product against microbial proliferation but should not compromise product performance. In practice, this means that any preservative or antimicrobial agent must:
- Exert a wide spectrum of antimicrobial activity against all possible micro-organisms at low inclusion levels
- Maintain activity throughout product manufacture, shelf life and usage
- Be non-toxic and compatible with other constituents of the preparation
- Not compromise the quality or performance of product, pack or delivery system.
- Not adversely affect patient safety or tolerance of the product.
Criteria for Choosing Antimicrobial Preservatives Include:
- Preservative Dose
- Effects on the Active Ingredient
- Antimicrobial Functionality
The evaluation of preservatives has traditionally involved time-consuming tests –pharmacopoeial antimicrobial effectiveness tests (AET) or preservative efficacy tests (PET). Such tests involve challenging a product with a defined number of colony forming units (cfu) of a variety of test microorganisms (bacteria, yeasts and fungi), enumeration at time zero and then monitoring the kill / survival rate at defined time intervals up to 28-days. Test organisms that are recommended by all of the pharmacopoeias include:
- Gram positive coccus, Staphylococcus aureus.
- Gram negative rod, Pseudomonas aeruginosa.
- Fungi / mould, Aspergillus niger.
- Yeast, Candida albicans.
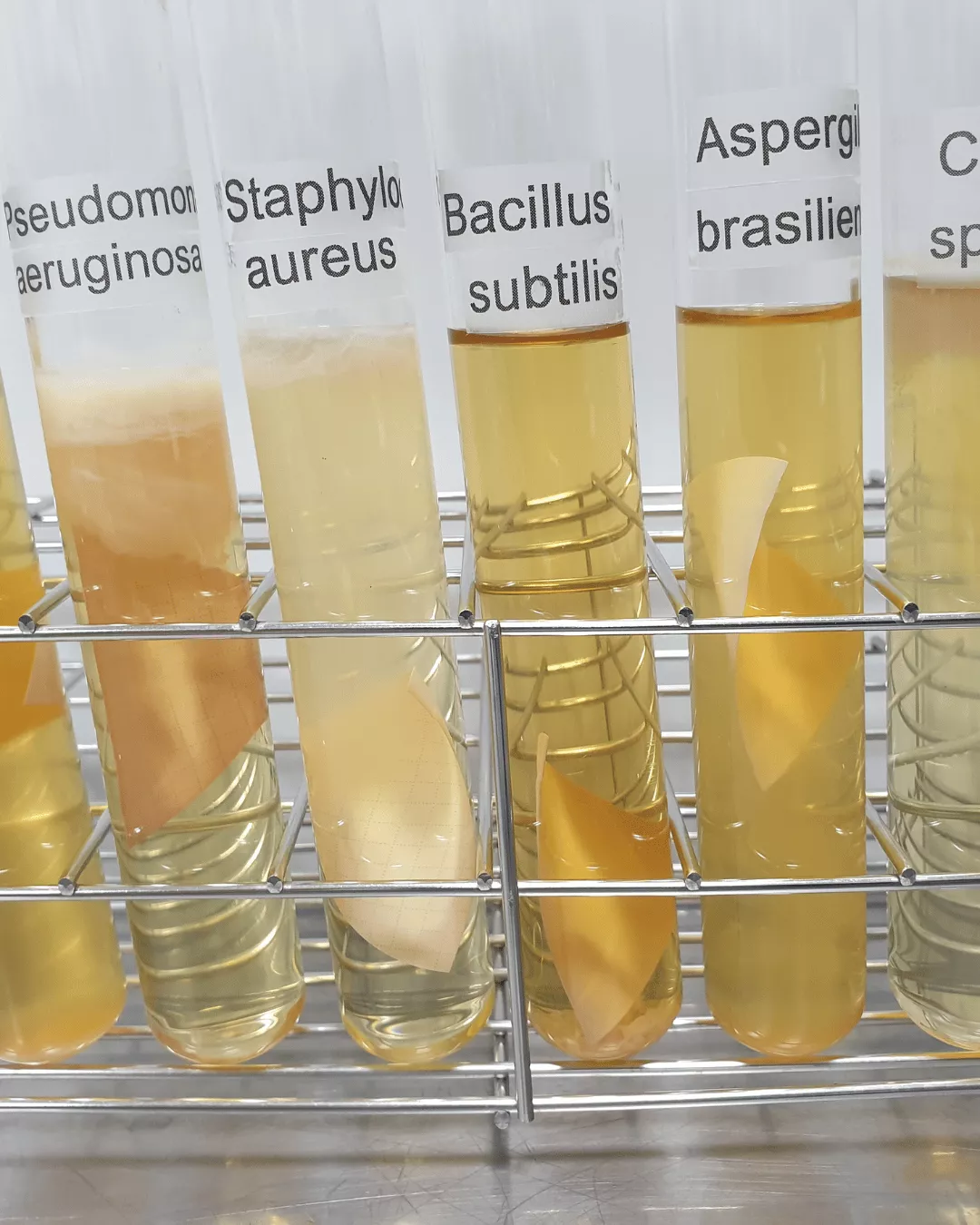
Preservative Efficacy Testing and Optimization
Regulatory guidelines, including USP and Ph. Eur. recommend the use of E. coli in preservative efficacy testing.
Additional organisms may be included depending on the specific process, facility, or material. These can include:
-
Burkholderia cepacia – an opportunistic pathogen often found in manufacturing settings
-
Bacillus subtilis – a spore-forming bacterium commonly used in contamination studies
Each challenge organism must show a satisfactory reduction in colony-forming units (CFUs), with no increase from the initial count after 14 and 28 days.
Testing Requirements
Preservative effectiveness must be confirmed at the end of the product’s shelf life. This ensures the preservative system remains effective throughout the product’s intended use. Some regulatory authorities also require confirmation of preservative performance during the in-use period (when the product is opened, used, and closed repeatedly) since this is when microbial exposure is highest.

New Testing Methods
Researchers are exploring faster, more efficient alternatives to traditional pharmacopoeial tests. These include:
-
ATP bioluminescence
-
Electrical impedance spectroscopy
-
Spectrofluorimetry
-
Chemiluminescence
These techniques could enable automated, high-throughput screening during product development.
Sign Up to Our Newsletter
Common Antimicrobial Preservatives:
- Phenol and Benzyl alcohol – effective for peptide and protein products
- Phenoxyethanol – vaccine preservation
A combination of propylparaben and methylparaben or benzyl alcohol is usually present in small volume parenteral formulations. It is required especially for multi-dose or semi-solid parenteral formulations.
For a preservative to be effective, it must decrease the percentage of the microbes and prevent any re-growth and these effects must be both microbiostatic and microbiocidal in nature. A microbicidal agent kills microorganisms such as bacteria, while a microbiostatic agent only prohibits the growth of such microorganisms. In the presence of microbiostatics, the microorganisms eventually die due to lack of reproduction. Microbicidals are irreversible and lethal, while microbiostatics are reversible.
Synergizing of Preservatives
Some preservatives don’t work well against certain microbes. To be effective, they often need to be combined with others.
For example, benzalkonium chloride is not effective against some strains of Pseudomonas aeruginosa. This bacterium is a common cause of hospital-acquired infections and is known for its resistance to antibiotics—even as early as 2015.
About Pseudomonas aeruginosa
-
Found in hospitals and healthcare settings
-
Can infect the lungs, bones, joints, skin, blood, and urinary tract
-
Especially dangerous for people with weak immune systems
-
Often treated with a combination of antibiotics (like a beta-lactam and an aminoglycoside), especially in serious cases such as sepsis or neutropenia
Combining certain ingredients can boost activity against Pseudomonas aeruginosa:
-
Benzyl alcohol, EDTA, and 2-phenylethanol improve its effectiveness
-
Organo-mercurials, cetrimide, chlorhexidine, and 3-cresol also show synergy when used together
These combinations help extend the antimicrobial coverage and reduce the risk of resistance.
Preservative Concentrations Recommended for Parenteral Preparation
| Sr. No. | Name | Recommended Concentration |
|---|---|---|
| 1. | Benzyl Alcohol | 0.5 to 10% |
| 2. | Benzalkonium Chloride | 0.01% |
| 3. | Butyl Paraben | 0.015% |
| 4. | Chlorobutanol | 0.25 to 0.5% |
| 5. | Meta Cresol | 01 to 0.25% |
| 6. | Chlorocresol | 0.1 to 0.18% |
| 7. | Methyl Paraben | 0.01 to 0.5% |
| 8. | Phenyl Ethyl Alcohol | 0.25 to 0.002% |
| 9. | Propyl Paraben | 0.005 to 0.002% |
| 10. | Phenol | 0.065 to 0.02% |
Preservative Concentration for Liquid Oral Preparation
| Sr. No. | Name | Recommended Concentration |
|---|---|---|
| 1. | Benzonic Acid | 0.1 to 0.2% |
| 2. | Sorbic Acid | 0.1 to 0.2% |
| 3. | Methyl Paraben | 0.25% |
| 4. | Propyl Paraben | 0.5 to 0.25% |
| 5. | Sodium Benzonate | 0.1 to 0.2% |
| 6. | Bronidol | 0.001 to 0.05% |
| 7. | Propylene Glycol | 0.25% |
If you would like any more information on this topic we are happy to try and help. While this topic is not a specialised area for us we do have a basic understanding and are more than happy to try and point you in the right direction for further assistance.
At LFA Machines, we pride ourselves on helping our customers set up and run effectively their production facilities for a wide range of products. We have worked with clients in the food, pharmaceutical, nutraceutical, holistic, chemical and industrial sectors to produce a wide range of tablets using our tablet presses. If you need any help getting set up please do get in contact and we will be happy to help.







